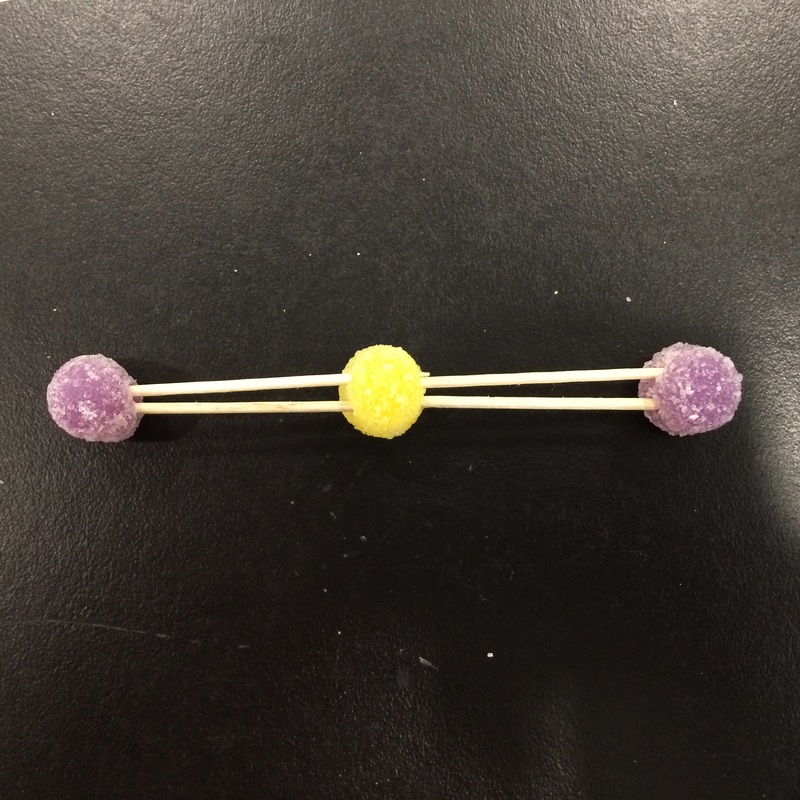
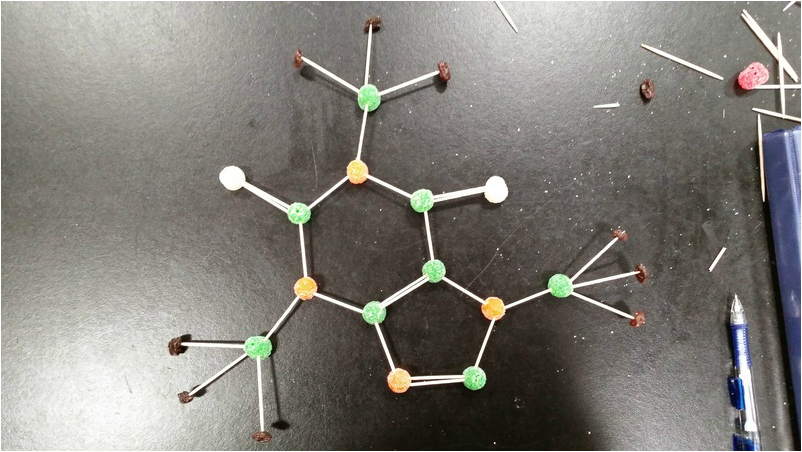
For this class lab, we were given a pile of gumdrops, a pile of toothpicks, and a box of raisins. The gumdrops were suppose to represent the atoms. The raisins were suppose to represent the hydrogen atoms. The toothpicks represented the bonds that bonded all the atoms together. We were suppose to put everything together in order to create different molecular structures. We had to make at least ten inorganic molecular structures from a table of formulas. And we were also supposed to make at least three organic molecular structures.
Here are the different molecular structures we made:
Phosphorus Pentafluoride - PF5
Here are the different molecular structures we made:
Phosphorus Pentafluoride - PF5
Phosphine - PH3
Aluminium Hydride - AlH3
Sulfur Dioxide - SO2
Formaldehyde - CH2O
Sulfur Trioxide - SO3
Aspirin - C9H8O4
Carbon Disulfide - CS2
Nicotine - C10H14N2
Ozone - O3
Nitrogen Trifluoride - NF3
Hydrogen Sulfide - H2S
Caffeine - C8H10N4O2
Before we started this lab, we learned the differences between an ionic compound and a covalent compound. In an ionic compound, the atoms turn into ions, which means they are either positively or negatively charged. In a covalent compound, the atoms share electrons with each other. I had trouble understanding these concepts before, so I am thankful we had this lab to clear everything up for me.
During this lab, we learned a lot about how molecular structure were supposed to work. Using the foods and toothpicks, we learned how exactly we were supposed to created molecular structures from looking at a chemical formula. To be more specific, I learned how each vertex in a molecular structure is suppose to represent a carbon atom. When there are six carbon atoms, they can make a ring of carbon. When there are two lines in the molecular structure, it is called a double bond. Since we did this lab, I finally had an understanding of how molecular structures work.
For completing this lab, I can come to the conclusion that some molecular structures are similar to other structures. They have the same structures but have different atoms in the positions. For example, aluminum hydride (AlH3) has the same structure as phosphine (PH3). Both structures have bonds with three hydrogen atoms. The only difference is that the main atoms, the Aluminum and Phosphorus, are different atoms.
During this lab, we learned a lot about how molecular structure were supposed to work. Using the foods and toothpicks, we learned how exactly we were supposed to created molecular structures from looking at a chemical formula. To be more specific, I learned how each vertex in a molecular structure is suppose to represent a carbon atom. When there are six carbon atoms, they can make a ring of carbon. When there are two lines in the molecular structure, it is called a double bond. Since we did this lab, I finally had an understanding of how molecular structures work.
For completing this lab, I can come to the conclusion that some molecular structures are similar to other structures. They have the same structures but have different atoms in the positions. For example, aluminum hydride (AlH3) has the same structure as phosphine (PH3). Both structures have bonds with three hydrogen atoms. The only difference is that the main atoms, the Aluminum and Phosphorus, are different atoms.



















 RSS Feed
RSS Feed
