The point of this lab was to create a battery made out of cardboard and pennies. These batteries were suppose to be able to power an LED.
The first thing we did was gather bunch of pennies that were made after 1982. We needed these pennies because they are 97.5 percent zinc and are only covered in copper.
We used sand paper to remove copper from one side of each penny. This revealed the zinc that was inside the copper coating. However, we decided to use zinc washers instead of sanding the copper off the pennies.
While we did this, we cut out penny sized circles from thin cardboard. We also soaked each of the cardboard circles in vinegar. The vinegar contained the electrolytes.
The first thing we did was gather bunch of pennies that were made after 1982. We needed these pennies because they are 97.5 percent zinc and are only covered in copper.
We used sand paper to remove copper from one side of each penny. This revealed the zinc that was inside the copper coating. However, we decided to use zinc washers instead of sanding the copper off the pennies.
While we did this, we cut out penny sized circles from thin cardboard. We also soaked each of the cardboard circles in vinegar. The vinegar contained the electrolytes.
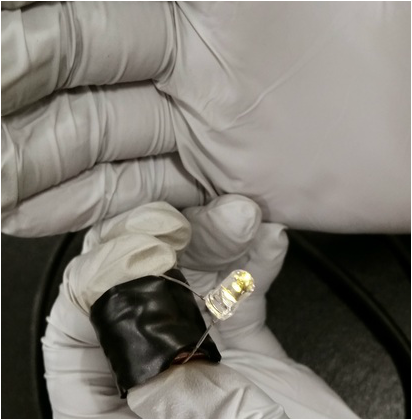
We then connect everything by stacking it. The order goes penny copper at the bottom, zinc washer in the middle, and vinegar cardboard at the top.
After we decided we had stacked on top of each other, we wrapped it in electrical tape. We cardboard was suppose to be the negative side, and the copper was the positive side.
We connected a LED to the battery to test if it worked. Our battery caused the LED to shine for only a few seconds. Then the battery stopped working.
We connected a LED to the battery to test if it worked. Our battery caused the LED to shine for only a few seconds. Then the battery stopped working.
The way I think this works is that the electrons flow in an upward direction between the two metals at the bottom and the cardboard with the electrolytes. When the LED was connected, the electrons would flow through the LED and cause it to shine. It is like a circuit where the positive leg of the LED is connected to the negative side of the battery and the negative leg with positive side.
Our group did not get any desirable results. We could only get the LED to shine for a few second. Even though we tried to rebuild it again, it still continued not to work. I believe this might be because our cardboard circle were touch each other, which messed up the entire battery.
I thought that I worked really well on this lab despite us failing to get it to work. I tried my best to think of solution as to why it did not work. I also worked hard to prepare each material by helping cut the cardboard into circle and stack everything together. What is really important is that I put my effort into this lab, which I did.
I worked with Angel and Franky for this lab. We have already worked together on plenty of lab, so we already know what to expect from each other. We were also able to communicate with each other, which meant that we did not argue. Despite failing, we all tried our best to get everything to work.
If I were to do this lab again, I would want to actually do everything correctly. I would want the LED to shine longer than just a few seconds. I would think it would be interesting if I created a larger stack, and see how much it would power things. There are really no ways for us to continue this lab besides actually getting it to work. To be honest I would not really want to continue this lab. I am just too discouraged by my failure the first time, I do not want to be hurt again.
For this lab, I learned more about how exactly batteries worked. This lab also helped me improve my skills on problem solving. This is because I was trying to find solutions as to why our battery was not working.
Our group did not get any desirable results. We could only get the LED to shine for a few second. Even though we tried to rebuild it again, it still continued not to work. I believe this might be because our cardboard circle were touch each other, which messed up the entire battery.
I thought that I worked really well on this lab despite us failing to get it to work. I tried my best to think of solution as to why it did not work. I also worked hard to prepare each material by helping cut the cardboard into circle and stack everything together. What is really important is that I put my effort into this lab, which I did.
I worked with Angel and Franky for this lab. We have already worked together on plenty of lab, so we already know what to expect from each other. We were also able to communicate with each other, which meant that we did not argue. Despite failing, we all tried our best to get everything to work.
If I were to do this lab again, I would want to actually do everything correctly. I would want the LED to shine longer than just a few seconds. I would think it would be interesting if I created a larger stack, and see how much it would power things. There are really no ways for us to continue this lab besides actually getting it to work. To be honest I would not really want to continue this lab. I am just too discouraged by my failure the first time, I do not want to be hurt again.
For this lab, I learned more about how exactly batteries worked. This lab also helped me improve my skills on problem solving. This is because I was trying to find solutions as to why our battery was not working.


 RSS Feed
RSS Feed
